Validation implications
Stage I validation requires a discussion of process knowledge and suggests the demonstration of a design space (2).
Submission implications
How the design space is used will modify how the submission will be generated and communicated. If it is used to show process knowledge, that is one kind of submission. If it is a basis for control with adjustments, that will follow another type of submission and is more complicated (see Figure 3).
Conclusion
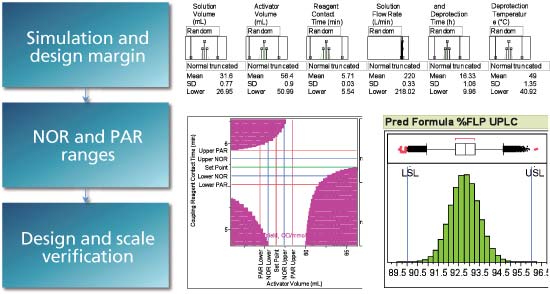
Modern ICH standards encourage design space generation in new product development. Use of design space increases product and process knowledge and reduces risk. Using a risk-based and multivariate approach is a best practice in generating the design space. Use of design space is best when establishing set points, tolerances, and PAR ranges. Best practices include determination of failure rates and design margin during development as well as using the transfer functions from the design space for process control. Intended design space and use should be clearly communicated to regulators. FDA generally welcomes discussion on design space with applicants so be sure to discuss the design space and submission logic with FDA working groups as needed.
References
1. ICH, Q8(R2) Pharmaceutical Development (2009).
2. FDA, Guidance for Industry, Process Validation: General Principles
and Practices (2011).
3. S. Chatterjee, “Design Space Considerations,” FDA, AAPS Annual Meeting, October 14, 2012, Chicago, ONDQA/CDER/FDA.
4. ICH, Q9 Quality Risk Management (2006).
About the author
Thomas A. Little, PhD, is president of Thomas A. Little Consulting, [email protected].