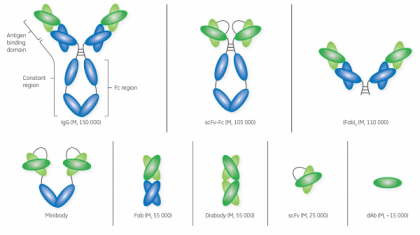
 In recent years, the global biopharma industry has seen a rapidly advancing trend toward biotherapeutics or ‘biologic’ drugs—drugs that are produced from living organisms or contain components of living organisms. In 2018 alone, the U.S. FDA approved 17 biologics out of a total of 59 drugs—the highest number to date.
In recent years, the global biopharma industry has seen a rapidly advancing trend toward biotherapeutics or ‘biologic’ drugs—drugs that are produced from living organisms or contain components of living organisms. In 2018 alone, the U.S. FDA approved 17 biologics out of a total of 59 drugs—the highest number to date.
This article will delve into why the pharma industry is heading toward a biologics revolution, what challenges exist when developing these drugs to be efficacious yet safe, and how novel analytical techniques can help circumvent these issues and optimize development.
Click here to read the article.
